Inclusion of Patient-Reported Outcomes in Adolescent and Young Adult Phase III Therapeutic Trials: An Analysis of Cancer Clinical Trials Registered on ClinicalTrials.gov - Value in Health

Overview of phase IV clinical trials for postmarket drug safety surveillance: a status report from the ClinicalTrials.gov registry | BMJ Open

U.S. GRADE Network blog: Registration of Trials Included in Systematic Reviews Has Improved Over Time, but Remains Under 50% for Most Years

Peer reviewed evaluation of registered end-points of randomised trials (the PRE-REPORT study): protocol for a stepped-wedge, cluster-randomised trial | BMJ Open
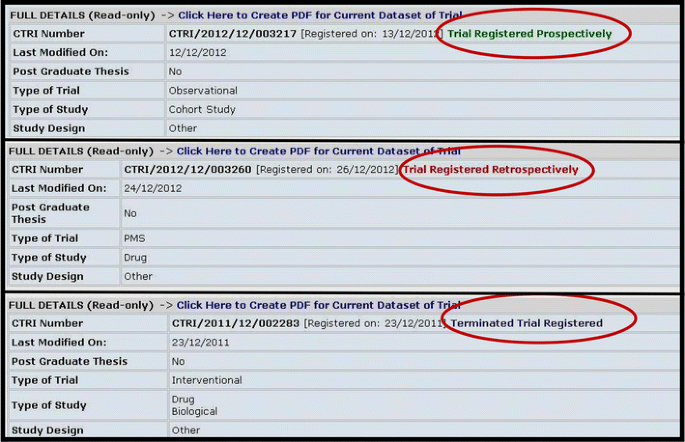
Challenges in Administering a Clinical Trials Registry: Lessons from the Clinical Trials Registry-India | SpringerLink
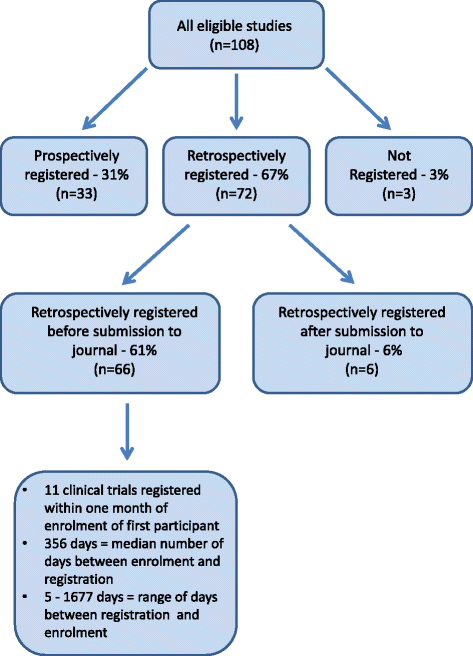
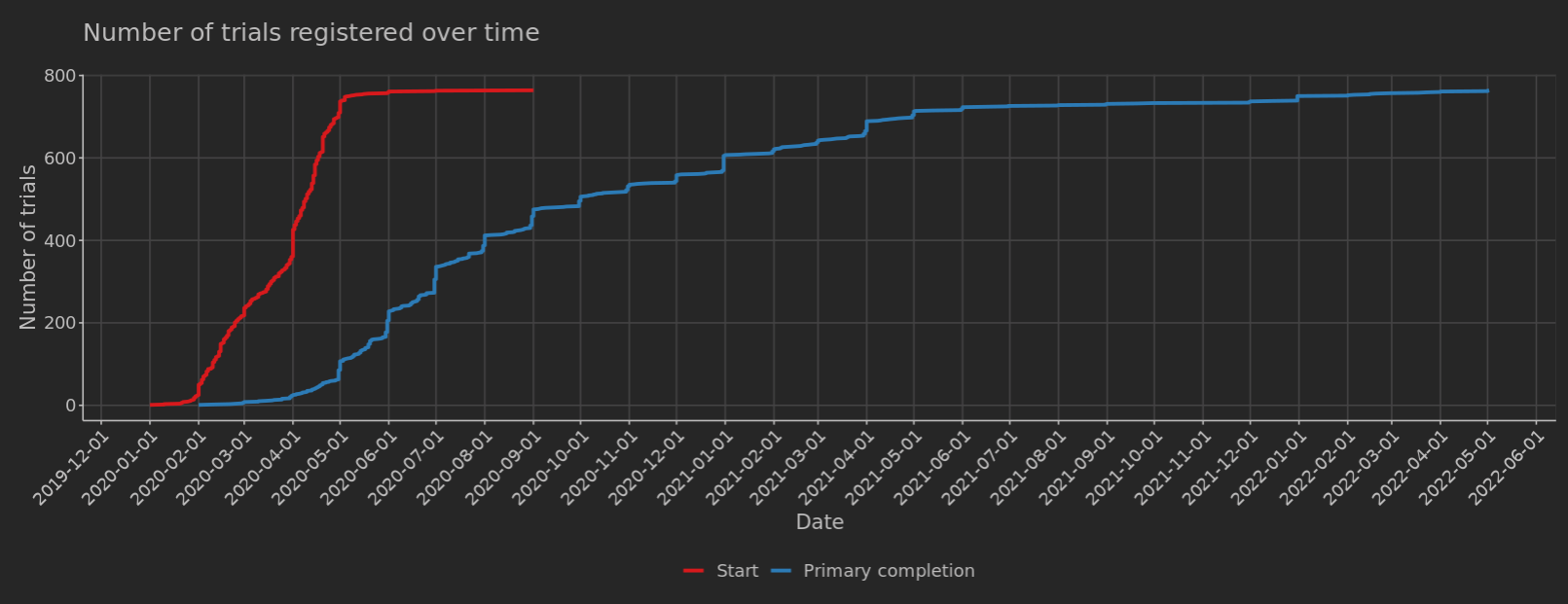
When are clinical trials registered? An analysis of prospective versus retrospective registration | Trials | Full Text

Is This Clinical Trial Fully Registered? — A Statement from the International Committee of Medical Journal Editors | NEJM

Number of registered trials by mapped drug name. Only the top 30 drugs... | Download Scientific Diagram