![14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/14221-01-3.png)
14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

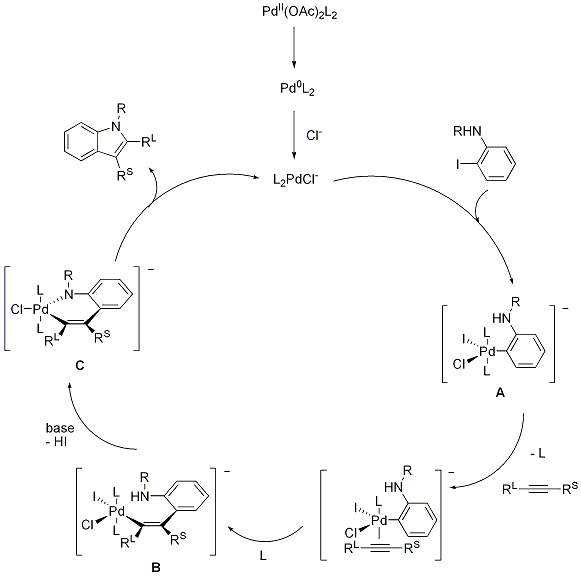
Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
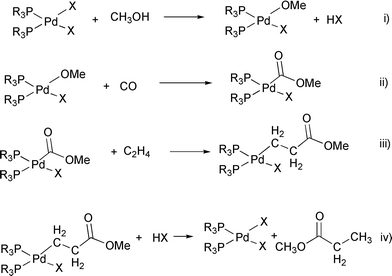
Evidence for the hydride mechanism in the methoxycarbonylation of ethene catalysed by palladium–triphenylphosphine complexes - Journal of the Chemical Society, Dalton Transactions (RSC Publishing)
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
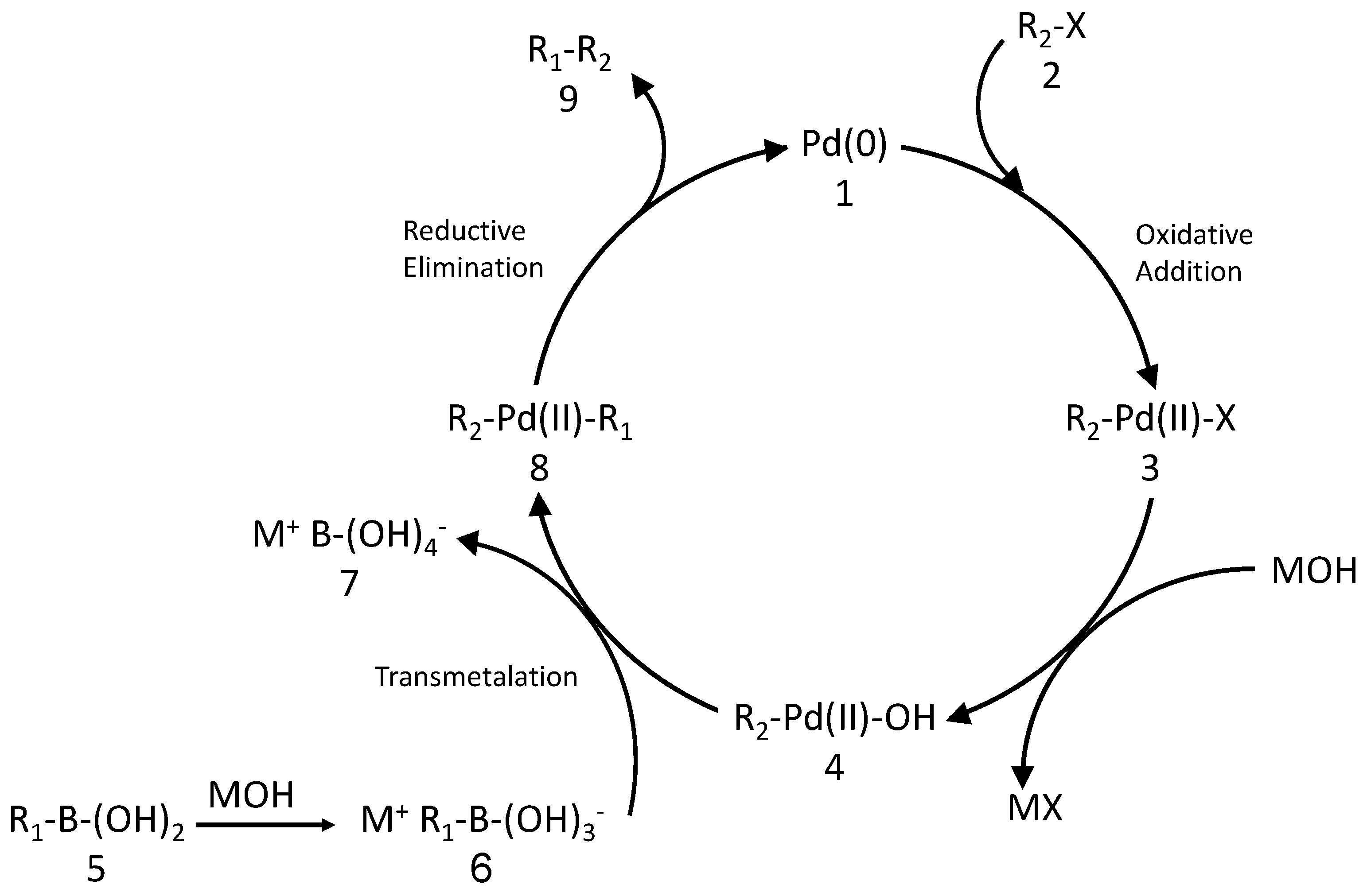
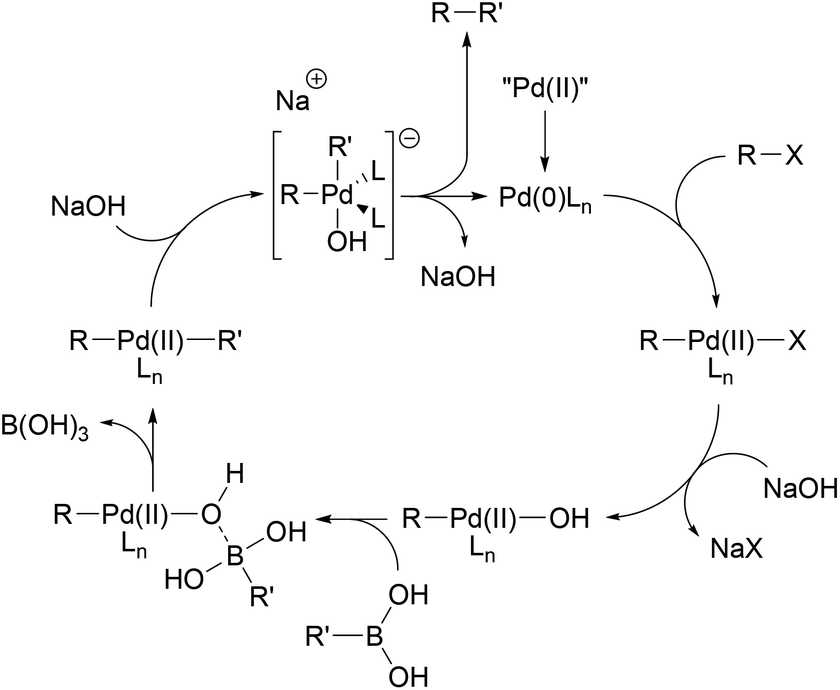
Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML

The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added Phosphorus Ligand as a “Green” Protocol: A Quantum Chemical Study on the Mechanism - Keglevich - 2017 - Advanced Synthesis & Catalysis -

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
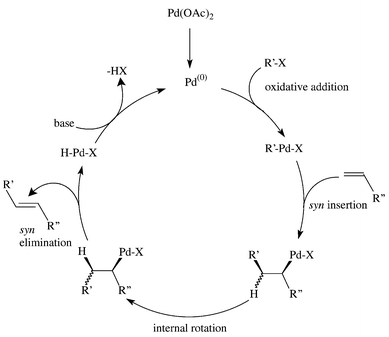
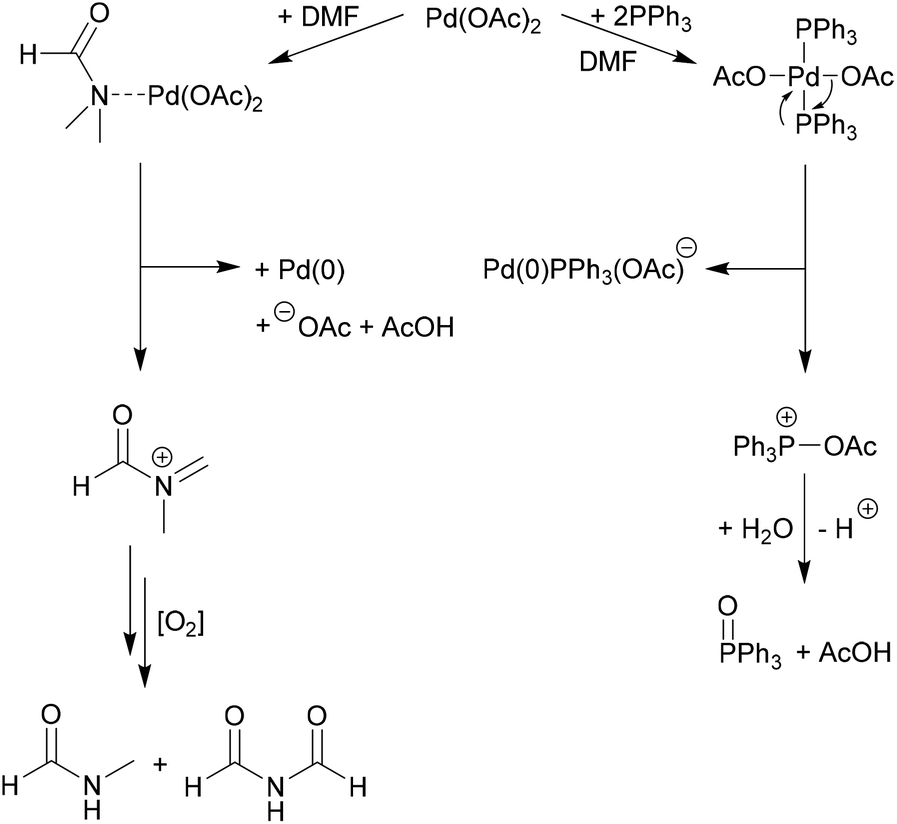
Structural characterisation of solution species implicated in the palladium-catalysed Heck reaction by Pd K-edge X-ray absorption spectroscopy: palladium acetate as a catalyst precursor - Journal of the Chemical Society, Dalton Transactions (RSC
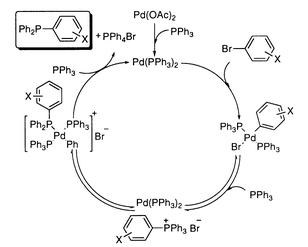
A general synthesis of aryl phosphines by palladium catalyzed phosphination of aryl bromides using triarylphosphines - Chemical Communications (RSC Publishing) DOI:10.1039/B002656P

The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added Phosphorus Ligand as a “Green” Protocol: A Quantum Chemical Study on the Mechanism - Keglevich - 2017 - Advanced Synthesis & Catalysis -
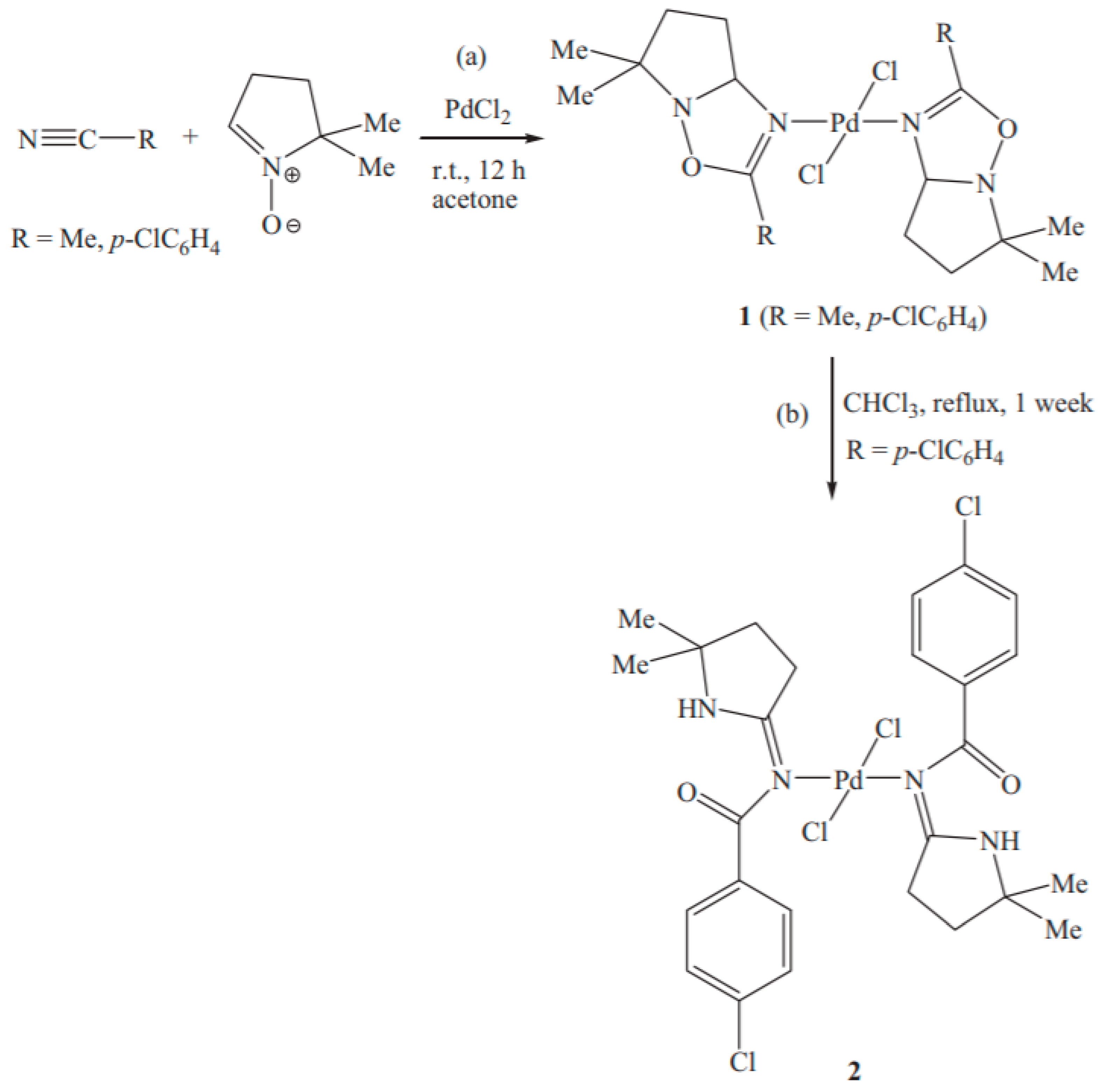
Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
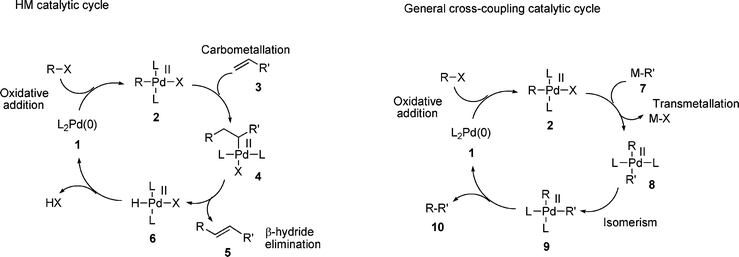
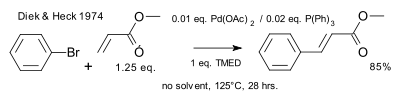
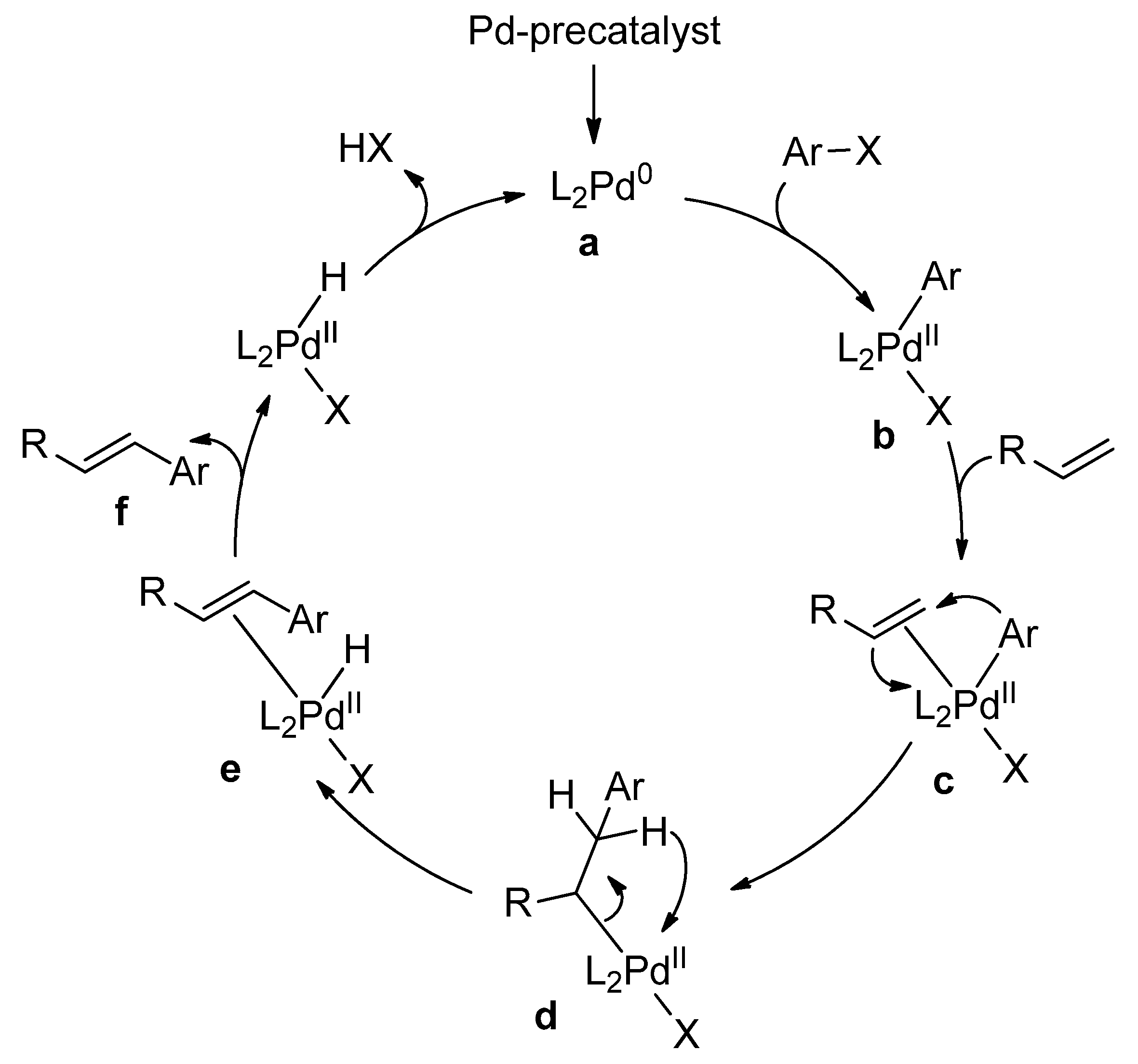
The Heck–Mizoroki cross-coupling reaction: a mechanistic perspective - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B611547K